Simplify compliance management in a secure, structured, validated environment
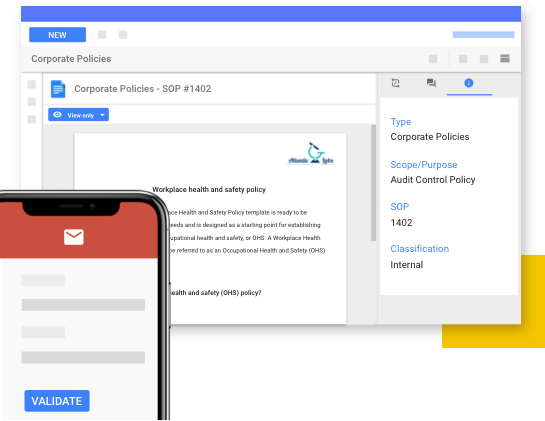
Comply with industry and corporate regulations such as GxP, FDA 21 CFR Part 11, and ISO 9001 via SaaS-based controlled and validated QMS options.
Eliminate time-consuming manual QMS processes that rely on paper or on-premise legacy systems, and boost efficiency for your organization’s users.
Reduce time-to-market and focus resources on innovation with speedy implementation and low ongoing maintenance of the AODocs QMS.

Our easy-to-deploy QMS solutions are available for Life Sciences and Manufacturing.

We had a pretty large wish list and we were able to hit it all. Everything was customized to fit exactly what we needed.
Larissa Pepper
HR Coordinator

AODocs has improved the productivity of the team by nearly 100% while also reducing the risk of costly human errors.
Decio Francini
IT Manager





